Defective gene expression has being recognized as the main causative agent or a key factor risk of a number of rare diseases,among which several ones affecting the central nervous system (CNS). It may be due to chromosomal microdeletions, spanning one or a few contiguous genes. Alternatively, the number of alleles may be correct, however these alleles may be poorly expressed, because of anomalous cis-active signals modulating transcription, intrinsic mRNA instability and/or poor translatability. Until now, pathologies linked to defective gene expression have been hardly tractable and, presently, no feasible-scalable approach is available for their therapy.
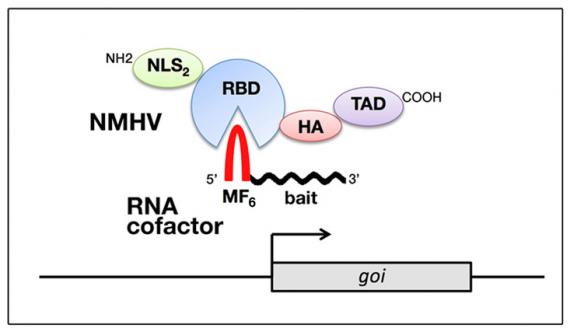
We are interested in developing artificial ncRNA-based devices for stimulating transcription of endogenous genes ad libitum, as key scalable tools for rationale treatment of haploinsufficiencies and other defective gene expression pathologies.
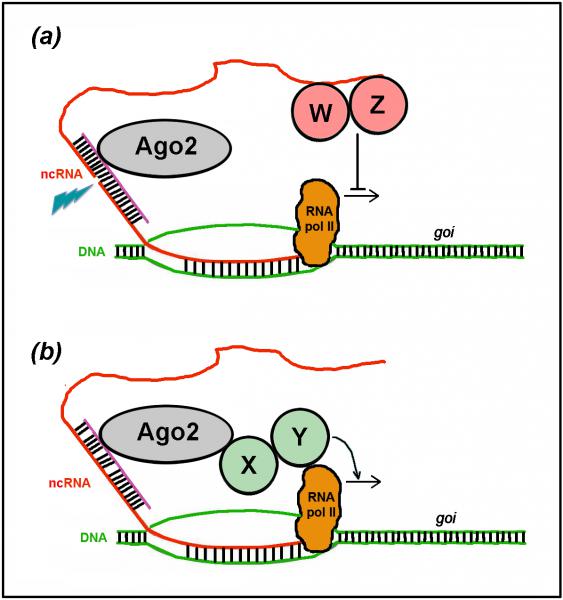
Among RNA-based devices promoting transcription of endogenous genes there are small activating RNAs (saRNAs). These are promoter-targeted, miRNA/siRNA-like molecules, supposed to (a) destabilize transcription-inhibiting ncRNAs or (b) drive transcription-promoting complexes to chromatin. The transcription gain they elicit is small, however it may be sufficient to impact the macroscopic behaviour of cells in a robust and predictable way. Moreover, silent genes generally do not respond to them. As such, saRNAs are a promising tool for clean therapeutic stimulation of gene transcritpion. We selected a number of saRNAs upregulating haploinsufficient genes implicated in CNS morphogenesis and physiology. We are studying their mechanism of action. We are working at their exploitation for in vivo correction of gene haploinsufficiency.